In an increasingly digital healthcare landscape, electronic product information (ePI) is poised to become the standard for medication information across the EU in the coming years. ePI involves converting traditional, unstructured product documents—such as Word or PDF files—into structured FHIR XML or JSON formats. This transformation aims to make prescribing information, labeling, and patient leaflets more accessible, interoperable, and easily integrated into digital healthcare systems. (electronic-product-information-epi-roadmap_en)
Every software vendor, healthcare institution and pharmacy that displays medicine information will be legally required to switch to the digital ePI standard (XML/FHIR).
Important information on behalf of Leaphy, a digital health technology company specialised in enabling technical compliance with the EU electronic Product Information (ePI) Common Standard and IDMP/SPOR frameworks.
As the EU moves toward mandatory ePI implementation (2026–2027), many Regulatory Information Management (RIM) and IDMP platforms are now looking for reliable, standards-aligned components that can transform approved product information into machine-readable, accessible, and resolvable ePI.
Leaphy’s platform is designed precisely for this:
• PDF → XML/FHIR conversion aligned to EMA/HMA Common ePI Standard and SPOR/IDMP master data.
• Automated validation engine ensuring conformance with the ePI schema and metadata rules.
• Repository and version management with audit trail and approval workflows.
• Resolver service connecting GTINs, NTINs, and national codes from the existing GS1 DataMatrix 2D code on packs to the correct ePI endpoint — no packaging changes required.
• API and integration layer for embedding directly within RIM or submission systems.
We see strong potential for collaboration — either as an OEM integration, API partnership, or co-marketed compliance module that enhances your RIM suite’s ePI readiness.
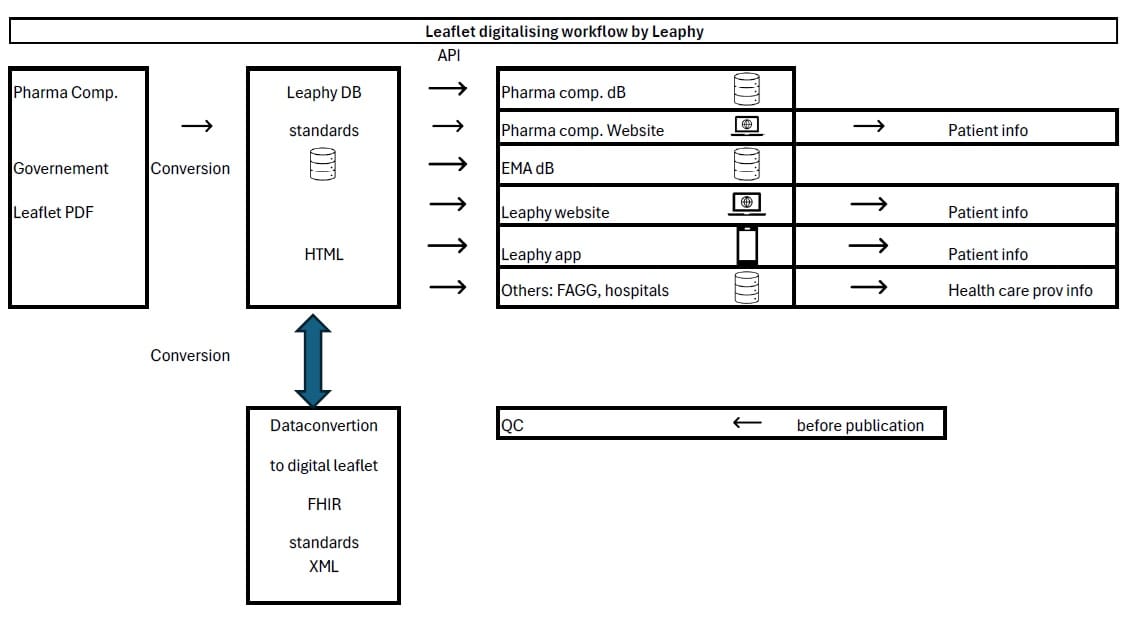
• See also dataflow below
Please do not hesitate to contact us with the contact form for any help on digitalisation of your leaflets and importing them into the EMA database.
Leaphy has Regulatory and Technical Expertise since many years